Lancelet
| Lancelets | |
|---|---|
 | |
| Lancelet (Branchiostoma lanceolatum) | |
| Scientific classification | |
| Kingdom: | Animalia |
| Phylum: | Chordata |
| Subphylum: | Cephalochordata Haeckel 1866 |
| Class: | Leptocardii Müller, 1845 |
| Order: | Amphoxiformes |
| Families | |
| Synonyms | |
|
Branchiostomiformes[1] | |
The lancelets (from "lancet") — also known as amphioxi (singular, amphioxus) — comprise some 32 species[2] of fish-like marine chordates in the order Amphioxiformes, with a global distribution in shallow temperate (as far north as Scotland)[3] and tropical seas, usually found half-buried in sand. They are the modern representatives of the subphylum Cephalochordata, formerly thought to be the sister group of the craniates.[4] In Asia, they are harvested commercially as food for humans and domesticated animals. They are an important object of study in zoology as they provide indications about the evolutionary origins of the vertebrates. Lancelets serve as an intriguing comparison point for tracing how vertebrates have evolved and adapted. Although lancelets split from vertebrates more than 520 million years ago, their genomes hold clues about evolution, particularly how vertebrates have employed old genes for new functions.[5] They are regarded as similar to the archetypal vertebrate form.
The first representative organism of the group to be described was Branchiostoma lanceolatum. It was described by Peter Simon Pallas in 1774 as molluscan slugs in the genus Limax. It was not until 1834 that Gabriel Costa brought the phylogenetic position of the group closer to the agnathan vertebrates (hagfish and lampreys), including it in the new genus Branchiostoma (from the Greek, branchio = "gills", stoma = "mouth").[6] In 1836, Yarrel renamed the genus as Amphioxus (from the Greek: "pointed on both sides"), now considered an obsolete synonym of the genus Branchiostoma. Today, the term "amphioxus" is still used as a common name for the Amphioxiformes, along with "lancelet", especially in the English language. A non-technical review of all aspects of lancelet biology is: Stokes, M. D. and Holland, N. D. 1998. American Scientist 86: 552-560. The genome of the Florida lancelet (Branchiostoma floridae) has been sequenced.[7]
Anatomy
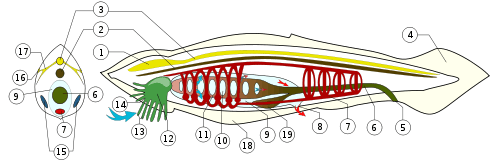

A: buccal cirri, B: wheel organ, C: velum, D: rostrum, E: notochord extending beyond nerve cord, F: nerve cord, G: Hatschek's pit, H: fin rays, I: gill bar, J: buccal cavity (vestibule)
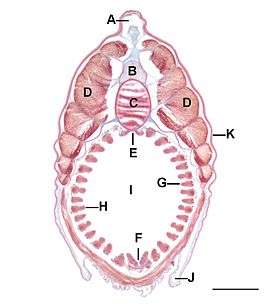
A: fin box, B: nerve cord, C: notochord,
D: myomeres, E: epibranchial groove,
F: endostyle, G: secondary gill bar,
H: primary gill bar, I: pharyngeal cavity,
J: metapleural fold, K: epidermis.
Lancelets are typically 5 centimetres (2.0 in) long, or 7 centimetres (2.8 in) at the longest. They have a translucent, somewhat fish-like body, but without any paired fins or other limbs. A relatively poorly developed tail fin is present, so they are not especially good swimmers. While they do possess some cartilage-like material stiffening the gill slits, mouth, and tail, they have no true skeleton.[8]
Nervous system
In common with vertebrates, lancelets have a hollow nerve cord running along the back, pharyngeal slits and a tail that runs past the anus. Also like vertebrates, the muscles are arranged in blocks called myomeres.
Unlike vertebrates, the dorsal nerve cord is not protected by bone but by a simpler notochord made up of a cylinder of cells that are closely packed to form a toughened rod. The lancelet notochord, unlike the vertebrate spine, extends into the head. This gives the subphylum its name (cephalo- meaning 'relating to the head'). The nerve cord is only slightly larger in the head region than in the rest of the body, so that lancelets do not appear to possess a true brain. However, developmental gene expression and Transmission electron microscopy indicate the presence of a diencephalic forebrain, a possible midbrain, and a hindbrain.[9] [10]
Visual system
Lancelets have two known kinds of light-sensing structures, Joseph cells, and Hesse organs, as well as a "frontal eye" and lamellar body. The frontal eye is widely thought to be light-sensitive, although this has not been confirmed by electrophysiological measurement. The lamellar body, a possible homologue of the pineal body, was once thought to be light-sensitive, although this has been called into question. All of these organs and structures are located in the neural tube, with the frontal eye at the front, followed by the lamellar body, the Joseph cells, and the Hesse organs.[11] [12] [13]
Joseph cells and Hesse organs
Joseph cells are bare photoreceptors surrounded by a band of microvilli. These cells bear the opsin melanopsin. The Hesse organs (also known as dorsal ocelli) consist of a photoreceptor cell surrounded by a band of microvill and bearing melanopsin, but half enveloped by a cup-shaped pigment cell. The peak sensitivy of both cells is ~470 nm[14] (blue).
Both the Joseph cells and Hesse organs are in the neural tube, the Joseph cells forming a dorsal column, the Hesse organs in the ventral part along the length of the tube. The Joseph cells extend from the caudal end of the anterior vesicle (or cerebral vesicle) to the boundary between myomeres 3 and 4, where the Hesse organs begin and continue nearly to the tail.[15][16]
Frontal eye
The frontal eye consists of a pigment cup, a group of putative photoreceptor cells (termed Row 1), three rows of neurons (Rows 2-4), and glial cells. The frontal eye, which expresses the PAX6 gene, has been proposed as the homolog of vertebrate paired eyes, the pigment cup as the homolog of the RPE (retinal pigment epithelium), the putative photoreceptors as homologs of vertebrate rods and cones, and Row 2 neurons as homologs of the retinal ganglion cells.[17]
The pigment cup is oriented concave dorsally. Its cells contain the pigment melanin. [17][18]
The putative photoreceptor cells, Row 1, are arranged in two diagonal rows, one on either side of the pigment cup, symmetrically positioned with respect to the ventral midline. The cells are flask-shaped, with long, slender ciliary processes (one cilium per cell). The main bodies of the cells lie outside of the pigment cup, while the cilia extend into the pigment cup before turning and exiting. The cells bear the opsin c-opsin 1, except for a few which carry c-opsin 3.[17][19]
The Row 2 cells are serotonergic neurons in direct contact with Row 1 cells. Row 3 and 4 cells are also neurons. Cells of all four rows have axons that project into the left and right ventrolateral nerves. For Row 2 neurons, axon projections have been traced to the tegmental neuropil. The tegmental neuropil has been compared with locomotor control regions of the vertebrate hypothalamus, where paracrine release modulates locomotor patterns such as feeding and swimming.[17]
Digestive system
Lancelets also have oral cirri, thin tentacle-like strands that hang in front of the mouth and act as sensory devices and as a filter for the water passing into the body. Water passes from the mouth into the large pharynx, which is lined by numerous gill-slits. The ventral surface of the pharynx contains a groove, called the endostyle, which, connected to a structure known as Hatschek's pit, produces a film of mucus. Ciliary action pushes the mucus in a film over the surface of the gill slits, trapping suspended food particles as it does so. The mucus is collected in a second, dorsal, groove, and passed back to the rest of the digestive tract. Having passed through the gill slits, the water enters an atrium surrounding the pharynx, then exits the body via the atriopore.[8]
Both adults and larvae exhibit a "cough" reflex to clear the mouth or throat of debris or items too large to swallow. In larvae the action is mediated by the pharyngial muscles while in the adult animal it is accomplished by atrial contraction.[20][21]
The remainder of the digestive system consists of a simple tube running from the pharynx to the anus. The hepatic caecum, a single blind-ending caecum, branches off from the underside of the gut, with a lining able to phagocytize the food particles, a feature not found in vertebrates. Although it performs many functions of a liver, it is not considered a true liver but a homolog of the vertebrate liver.[22][23][24]
Other systems
Lancelets have no respiratory system, breathing solely through their skin, which consists of a simple epithelium. Despite the name, little if any respiration occurs in the gill slits, which are solely devoted to feeding. The circulatory system does resemble that of primitive fish in its general layout, but is much simpler, and does not include a heart. There are no blood cells, and no haemoglobin.[8]
The excretory system consists of segmented "kidneys" containing protonephridia instead of nephrons, and quite unlike those of vertebrates. Also unlike vertebrates, there are numerous, segmented gonads.[8]
Model organism
Lancelets became famous in the 1860s when Ernst Haeckel ("the German Darwin") began promoting them as a model for the ancestor of all vertebrates. By 1900 lancelets had become a model organism. By the mid-20th century they had fallen out of favor for a variety of reasons, including a decline of comparative anatomy and embryology, and due to the belief that lancelets were more derived than they appeared, e.g., the profound asymmetry in the larval stage.[25][26]
With the advent of molecular genetics lancelets are once again regarded as a model of vertebrate ancestors, and are in process of being revived as a model organism.[27][28]
Taxonomy
The Cephalochordata is traditionally seen as a sister subphylum to the vertebrates, with which it is grouped together into a clade (sometimes called Notochordata) which in turn is the sister group to the simpler still Urochordata. Newer research suggests this is not the case. The Cephalochordata is the most basal subphylum of the chordates,[31] while the sister group of the vertebrates are the tunicates, previously known as the urochordates.[7] However, older molecular studies[32] placed cephalochordates nearer to vertebrates, and "[m]ost authors regard amphioxus as the closest relative of the Vertebrata on the basis of 10–15 [morphological] features that are not seen in tunicates".[32]
The following are the species recognised by ITIS. Other sources, for instance Tudge,[26] show that there might be up to thirty species. [33]
- †Cathaymyrus Shu, Conway Morris & Zhang 1996
- †Cathaymyrus diadexus Shu, Conway Morris & Zhang 1996
- †Cathaymyrus haikouensis Luo & Hu 2001
- †Paleobranchiostoma hamatotergum Oelofsen & Loock 1981
- Family Asymmetronidae
- Genus Asymmetron Andrews 1893 [Amphioxides Gill 1895; Assymetron (sic) Kirkaldy 1895; Asymetron (sic) Zietz 1908]
- Asymmetron inferum Nishikawa 2004
- Asymmetron lucayanum Andrews 1893 (Sharptail lancelet) [Asymmetron caudatum Willey 1896; Asymmetron macricaudatum Parker 1904; Asymmetron orientale Parker 1904; Amphioxides stenurus Goldschmidt 1905; Amphioxides valdiviae Goldschmidt 1905; Branchiostoma pelagicum Gunther 1889; Branchiostoma valdiviae (Goldschmidt 1905); Epigonichthys lucayanum (Andrews 1893); Epigonichthys lucayanus (Andrews 1893)]
- Genus Epigonichthys Peters 1876 [Amphipleurichthys Whitley 1932; Bathyamphioxus Whitley 1932; Heteropleuron Kirkaldy 1895; Merscalpellus Whitley 1932; Notasymmetron Whitley 1932; Paramphioxus Haekel 1893; Zeamphioxus Whitley 1932]
- Epigonichthys australis (Raff 1912) [Asymetron australis Raffe 1912]
- Epigonichthys bassanus (Günther 1884) [Branchiostoma bassanum Gunther 1884]
- Epigonichthys cingalensis (Kirkaldy 1894); nomen dubium[34] [Heteropleuron cingalense Kirkaldy 1894]
- Epigonichthys cultellus Peters 1877 [Bathyamphioxus franzi Whitley 1932; Epigonichthys pulchellus Gunther 1880; Heteropleuron hedleyi Haswell 1908; Branchiostoma cultellus (Peters 1877)]
- Epigonichthys hectori (Benham 1901) (Hector’s lancelet) [Heteropleuron hectori Benham 1901]
- Epigonichthys maldivensis (Foster Cooper 1903) [Asymmetron maldivensis Forster Cooper 1903; Heteropleuron agassizii Parker 1904; Heteropleuron maldivense Forster Cooper 1903; Heteropleuron parvum Parker 1904]
- Genus Asymmetron Andrews 1893 [Amphioxides Gill 1895; Assymetron (sic) Kirkaldy 1895; Asymetron (sic) Zietz 1908]
- Family Branchiostomidae Bonaparte 1841
- Genus Branchiostoma Costa 1834 non Newport 1845 non Banks 1905 [Amphioxus Yarrell 1836; Amphioxys Agassiz 1846; Limax Pallas 1774 non Linnaeus 1758 non Férussac 1819 non Martyn 1784; Dolichorhynchus Willey 1901 non Mulk & Jairajpuri 1974; Dolichorynchus Willey 1901; Dolichorhamphus (sic) Hubbs 1922]
- Branchiostoma africae Hubbs 1927
- Branchiostoma arabiae Webb 1957
- Branchiostoma bazarutense Gilchrist 1923
- Branchiostoma belcheri Gray 1847 (Belcher's lancelet)
- Branchiostoma bennetti Boschung & Gunter 1966 (Mud lancelet)
- Branchiostoma bermudae Hubbs 1922
- Branchiostoma californiense Andrews 1893 (Californian lancelet)
- Branchiostoma capense Gilchrist 1902
- Branchiostoma caribaeum Sundevall 1853 (Caribbean Lancelet)
- Branchiostoma clonaseum
- Branchiostoma elongatum Sundevall 1852
- Branchiostoma floridae Hubbs 1922 (Florida Lancelet)
- Branchiostoma gambiense Webb 1958
- Branchiostoma indicum Willey 1901
- Branchiostoma japonicum Willey 1896 (Pacific lancelet)
- Branchiostoma lanceolatum Pallas 1774 (European Lancelet)
- Branchiostoma leonense Webb 1956
- Branchiostoma longirostrum Boschung 1983 (Shellhash lancelet)
- Branchiostoma malayanum Webb 1956
- Branchiostoma moretonense Kelly 1966; nomen dubium[35][36]
- Branchiostoma nigeriense Webb 1955
- Branchiostoma platae Hubbs 1922
- Branchiostoma senegalense Webb 1955
- Branchiostoma tattersalli Hubbs 1922
- Branchiostoma virginiae Hubbs 1922 (Virginian lancelet)
- Genus Branchiostoma Costa 1834 non Newport 1845 non Banks 1905 [Amphioxus Yarrell 1836; Amphioxys Agassiz 1846; Limax Pallas 1774 non Linnaeus 1758 non Férussac 1819 non Martyn 1784; Dolichorhynchus Willey 1901 non Mulk & Jairajpuri 1974; Dolichorynchus Willey 1901; Dolichorhamphus (sic) Hubbs 1922]
References
- ↑ Classification of Class: Leptocardii - Ocean Biogeographic Information System: Canadian Museum of Nature (OBIS Canada)
- ↑ "Cephalochordata: EoL species". Retrieved 2014-06-12.
- ↑ "Year of discovery". Scotland.gov.uk. 2011-12-29. Retrieved 2012-08-13.
- ↑ Webb, J. E. (1958). "The Ecology of Lagos Lagoon. III. The Life History of Branchiostoma nigeriense Webb". Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences. The Royal Society. 241 (683): 335–353. doi:10.2307/92531. JSTOR 92531.
- ↑ Worm-like Marine Animal Providing Fresh Clues About Human Evolution Newswise, Retrieved on July 8, 2008.
- ↑ Garcia‐Fernàndez, J., & Benito‐Gutiérrez, È. (2009). It's a long way from amphioxus: descendants of the earliest chordate. Bioessays, 31(6), 665-675, .
- 1 2 Putnam, N. H.; Butts, T.; Ferrier, D. E. K.; Furlong, R. F.; Hellsten, U.; Kawashima, T.; Robinson-Rechavi, M.; Shoguchi, E.; Terry, A.; Yu, J. K.; Benito-Gutiérrez, E. L.; Dubchak, I.; Garcia-Fernàndez, J.; Gibson-Brown, J. J.; Grigoriev, I. V.; Horton, A. C.; De Jong, P. J.; Jurka, J.; Kapitonov, V. V.; Kohara, Y.; Kuroki, Y.; Lindquist, E.; Lucas, S.; Osoegawa, K.; Pennacchio, L. A.; Salamov, A. A.; Satou, Y.; Sauka-Spengler, T.; Schmutz, J.; Shin-i, T. (Jun 2008). "The amphioxus genome and the evolution of the chordate karyotype". Nature. 453 (7198): 1064–1071. Bibcode:2008Natur.453.1064P. doi:10.1038/nature06967. ISSN 0028-0836. PMID 18563158.
- 1 2 3 4 Romer, Alfred Sherwood; Parsons, Thomas S. (1977). The Vertebrate Body. Philadelphia, PA: Holt-Saunders International. pp. 18–21. ISBN 0-03-910284-X.
- ↑ Candiani, Simona; Moronti, Luca; Ramoino, Paola; Schubert, Michael; Pestarino, Mario (2012). "A neurochemical map of the developing amphioxus nervous system". BMC Neuroscience. 13 (1): 59. doi:10.1186/1471-2202-13-59. ISSN 1471-2202.
- ↑ Holland, L. Z. (2015). "The origin and evolution of chordate nervous systems" (PDF). Philosophical Transactions of the Royal Society B: Biological Sciences. 370 (1684): 20150048–20150048. doi:10.1098/rstb.2015.0048. ISSN 0962-8436.
- ↑ Nieuwenhuys, Rudolf; ten Donkelaar, Hans J.; Charles Nicholson (14 November 2014). The Central Nervous System of Vertebrates. Springer. p. 371. ISBN 978-3-642-18262-4. Retrieved 25 November 2015.
- ↑ Wanninger, Andreas (11 August 2015). Evolutionary Developmental Biology of Invertebrates 6: Deuterostomia. Springer. pp. 108–9. ISBN 978-3-7091-1856-6. Retrieved 21 November 2015.
- ↑ Lamb, Trevor D. (2013). "Evolution of phototransduction, vertebrate photoreceptors and retina". Progress in Retinal and Eye Research. 36: 52–119. doi:10.1016/j.preteyeres.2013.06.001. ISSN 1350-9462.
- ↑ del Pilar Gomez, M.; Angueyra, J. M.; Nasi, E. (2009). "Light-transduction in melanopsin-expressing photoreceptors of Amphioxus". Proceedings of the National Academy of Sciences. 106 (22): 9081–9086. doi:10.1073/pnas.0900708106. ISSN 0027-8424.
- ↑ Le Douarin, Nicole Marthe; Dupin, Elisabeth (23 November 2013). Paul Trainor, ed. Neural Crest Cells: Evolution, Development and Disease. Academic Press. p. 10. ISBN 978-0-12-404586-6. Retrieved 25 November 2015.
- ↑ Wicht, Helmut; Lacalli, Thurston C (2005). "The nervous system of amphioxus: structure, development, and evolutionary significance". Canadian Journal of Zoology. 83 (1): 122–150. doi:10.1139/z04-163. ISSN 0008-4301.
- 1 2 3 4 Vopalensky, P.; Pergner, J.; Liegertova, M.; Benito-Gutierrez, E.; Arendt, D.; Kozmik, Z. (2012). "Molecular analysis of the amphioxus frontal eye unravels the evolutionary origin of the retina and pigment cells of the vertebrate eye" (PDF). Proceedings of the National Academy of Sciences. 109 (38): 15383–15388. doi:10.1073/pnas.1207580109. ISSN 0027-8424.
- ↑ Jankowski, Roger (19 March 2013). The Evo-Devo Origin of the Nose, Anterior Skull Base and Midface. Springer Science & Business Media. p. 152. ISBN 978-2-8178-0422-4. Retrieved 7 December 2015.
- ↑ Lacalli, T. C. (1996). "Frontal Eye Circuitry, Rostral Sensory Pathways and Brain Organization in Amphioxus Larvae: Evidence from 3D Reconstructions" (PDF). Philosophical Transactions of the Royal Society B: Biological Sciences. 351 (1337): 243–263. doi:10.1098/rstb.1996.0022. ISSN 0962-8436.
- ↑ Lesley J. Rogers; Richard Andrew (25 March 2002). Comparative Vertebrate Lateralization. Cambridge University Press. pp. 72–. ISBN 978-1-139-43747-9.
- ↑ Rigon, Francesca; Stach, Thomas; Caicci, Federico; Gasparini, Fabio; Burighel, Paolo; Manni, Lucia (2013). "Evolutionary diversification of secondary mechanoreceptor cells in tunicata". BMC Evolutionary Biology. 13 (1): 112. doi:10.1186/1471-2148-13-112. ISSN 1471-2148.
- ↑ Yuan, Shaochun; Ruan, Jie; Huang, Shengfeng; Chen, Shangwu; Xu, Anlong (2015). "Amphioxus as a model for investigating evolution of the vertebrate immune system" (PDF). Developmental & Comparative Immunology. 48 (2): 297–305. doi:10.1016/j.dci.2014.05.004. ISSN 0145-305X.
- ↑ Yu, Jr-Kai Sky; Lecroisey, Claire; Le Pétillon, Yann; Escriva, Hector; Lammert, Eckhard; Laudet, Vincent (2015). "Identification, Evolution and Expression of an Insulin-Like Peptide in the Cephalochordate Branchiostoma lanceolatum". PLOS ONE. 10 (3): e0119461. doi:10.1371/journal.pone.0119461. ISSN 1932-6203.
- ↑ Escriva, Hector; Chao, Yeqing; Fan, Chunxin; Liang, Yujun; Gao, Bei; Zhang, Shicui (2012). "A Novel Serpin with Antithrombin-Like Activity in Branchiostoma japonicum: Implications for the Presence of a Primitive Coagulation System". PLoS ONE. 7 (3): e32392. doi:10.1371/journal.pone.0032392. ISSN 1932-6203.
- ↑ Hopwood, Nick (2015). "The cult of amphioxus in German Darwinism; or, Our gelatinous ancestors in Naples' blue and balmy bay". History and Philosophy of the Life Sciences. 36 (3): 371–393. doi:10.1007/s40656-014-0034-x. ISSN 0391-9714.
- 1 2 Tudge, Colin (2000). The Variety of Life. Oxford University Press. ISBN 0198604262.
- ↑ Holland, L.Z.; Laudet, V.; Schubert, M. (2004). "The chordate amphioxus: an emerging model organism for developmental biology" (PDF). Cellular and Molecular Life Sciences. 61 (18). doi:10.1007/s00018-004-4075-2. ISSN 1420-682X.
- ↑ Garcia-Fernàndez, Jordi; Benito-Gutiérrez, Èlia (2009). "It's a long way from amphioxus: descendants of the earliest chordate". BioEssays. 31 (6): 665–675. doi:10.1002/bies.200800110. ISSN 0265-9247.
- ↑ Gewin, V. (2005). "Functional genomics thickens the biological plot". PLoS biology, 3 (6), e219. doi:10.1371/journal.pbio.0030219
- ↑ Lancelet (amphioxus) genome and the origin of vertebrates Ars Technica, 19 June 2008.
- ↑ Henry Gee (2008). "Evolutionary biology: The amphioxus unleashed". Nature 453: 999-1000.
- 1 2 Michael J. Benton (2005). Vertebrate Palaeontology, Third Edition 8. Oxford: Blackwell Publishing. ISBN 0-632-05637-1.
- ↑ WoRMS Editorial Board (2013). "World Register of Marine Species- Cephalochordates species list". Retrieved 2013-10-22.
- ↑ WoRMS: Epigonichthys
- ↑ URMO taxon details: Branchiostoma mortonense
- ↑ WoRMS: Branchiostoma mortonense
Further reading
- Stach, T. G. (2004). Cephalochordata (Lancelets). In M. Hutchins, R. W. Garrison, V. Geist, P. V. Loiselle, N. Schlager, M. C. McDade, ...W. E. Duellman (Eds.), Grzimek's Animal Life Encyclopedia (2nd ed., Vol. 1, pp. 485–493). Detroit: Gale.
External links
| Wikimedia Commons has media related to Amphioxiformes. |
| Wikispecies has information related to: Cephalochordata |
- Probable Lancelet’s ancestor at PENICHEFOSSIL
- Branchiostoma japonicum and B. belcheri are Distinct Lancelets (Cephalochordata) in Xiamen Waters in China – article at Research Gate
- Error in the Genealogy of Human
- A special issue of Amphioxus Research (I)
- A special issue of Amphioxus Research (II)
- Amphioxus and the T-box gene
- A movie of the amphioxus embryonic development on YouTube
- Scripps Scientists Discover Fluorescence in Key Marine Creature
- Amphioxus at GeoChemBio: taxonomy, brief facts, life cycle and embryology
- View the braFlo1 genome assembly in the UCSC Genome Browser.
