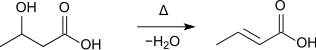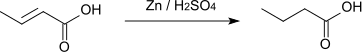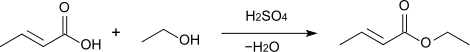Crotonic acid
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
(2E)-But-2-enoic acid | |
| Other names
(E)-But-2-enoic acid (E)-2-Butenoic acid Crotonic acid trans-2-Butenoic acid beta-Methylacrylic acid 3-Methylacrylic acid | |
| Identifiers | |
| 107-93-7 | |
| 3D model (Jmol) | Interactive image Interactive image |
| ChEBI | CHEBI:41131 |
| ChEMBL | ChEMBL1213528 |
| ChemSpider | 552744 |
| DrugBank | DB02074 |
| ECHA InfoCard | 100.003.213 |
| PubChem | 637090 |
| UNII | YW5WZZ4O5Q |
| |
| |
| Properties | |
| C4H6O2 | |
| Molar mass | 86.09 g·mol−1 |
| Density | 1.02 g/cm3 |
| Melting point | 70 to 73 °C (158 to 163 °F; 343 to 346 K) |
| Boiling point | 185 to 189 °C (365 to 372 °F; 458 to 462 K) |
| Acidity (pKa) | 4.69 [1] |
| Hazards | |
| Safety data sheet | SIRI.org |
| Related compounds | |
| Other anions |
crotonate |
| Related carboxylic acids |
propionic acid acrylic acid butyric acid succinic acid malic acid tartaric acid fumaric acid pentanoic acid |
| Related compounds |
butanol butyraldehyde crotonaldehyde 2-butanone |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Crotonic acid ((E)-2-butenoic acid) or is a short-chain unsaturated carboxylic acid, described by the formula CH3CH=CHCO2H. It is also called crotonic acid because it was erroneously thought to be a saponification product of croton oil.[2] It crystallizes as needles from hot water. The cis-isomer of crotonic acid is called isocrotonic acid.
Production
Crotonic acid acid may be obtained by several methods:
- by oxidation of (E)-2-butenal:[3]
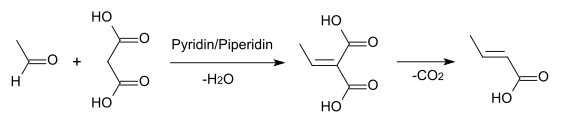
- by Knoevenagel condensation of acetaldehyde with propanedioic acid in pyridine:[4]
- or by alkaline hydrolysis of allyl cyanide after the intramolecular rearrangement of the double bond:[5][6]
- Furthermore, it is formed during the distillation of 3-hydroxybutanoic acid:[7]
Properties
Crotonic acid acid crystallizes in long needles or as large panels in the monoclinic crystal system in the space group P21/a (space group no. 14, position 3) with the lattice parameters a = 971 pm, b = 690 pm, c = 775 pm and β = 104.0°. The unit cell contains four formula units.[8] The compound is soluble in water and many organic solvents such as ethanol, acetone or toluene. Its odor is similar to butanoic acid and irritates eyes, skin and respiratory system.[9]
Reactions
Crotonic acid can be converted into butanoic acid by hydrogenation with zinc and sulfuric acid.[10]
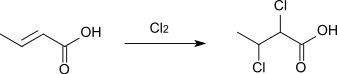
It forms with elemental chlorine or bromine 2,3-dihalogenbutanoic acids:[10]
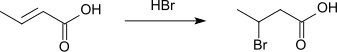
The electrophilic addition of hydrogen bromide forms 3-bromobutanoic acid.[10][11] The substitution pattern arises from the electron-withdrawing effect of the carboxyl group. The carbenium ion is more stable at position 3, this is where the bromine anion attaches.
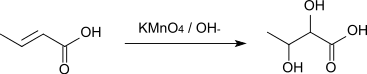
The reaction with alkaline potassium permanganate solution affords 2,3-dihydroxybutanedioic acid.[10]
Heating with acetic anhydride leads to (E)-2-butenoic-anhydride:[12]
The esterification of crotonic acid with methanol or ethanol using sulfuric acid as a catalyst provides the corresponding esters:
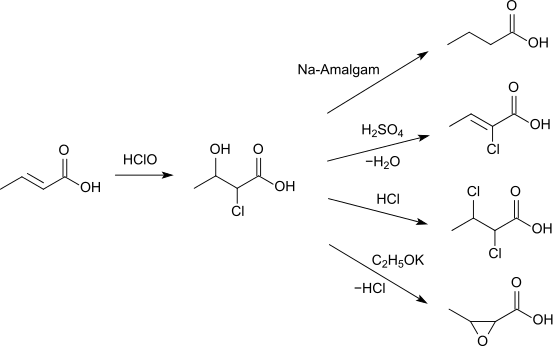
Butenoic reacts with hypochlorous acid to 2-chloro-3-hydroxybutanoic acid. This can either be reduced with sodium amalgam to butanoic acid, can form with sulfuric acid 2-chlorobutenoic acid, react with hydrogen chloride to 2,3-dichlorobutenoic acid or with potassium ethoxide to 3-methyloxirane-2- carboxylic acid.[13]
Use
Crotonic acid is used for the preparation of (racemic) DL-threonine by alpha-functionalization using mercury(II) acetate.[14] For the production of plastics, it may be copolymerized with vinyl acetate.[9]
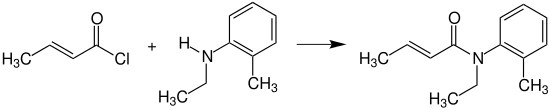
Crotonyl chloride reacts with N-ethyl-2-methylaniline (N-ethyl-o-toluidine) to provide crotamiton, which is used as an agent against scabies.[15]
See also
References
- ↑ Dawson, R. M. C., et al., Data for Biochemical Research, Oxford, Clarendon Press, 1959.
- ↑
 Chisholm, Hugh, ed. (1911). "Crotonic Acid". Encyclopædia Britannica. 7 (11th ed.). Cambridge University Press. p. 511.
Chisholm, Hugh, ed. (1911). "Crotonic Acid". Encyclopædia Britannica. 7 (11th ed.). Cambridge University Press. p. 511. - ↑ Hans Beyer und Wolfgang Walter: Organische Chemie, S. Hirzel Verlag, Stuttgart 1984, ISBN 3-7776-0406-2, S. 230.
- ↑ Hans Beyer und Wolfgang Walter: Organische Chemie, S. Hirzel Verlag, Stuttgart 1984, ISBN 3-7776-0406-2, S. 229–230.
- ↑ A. Rinne, B. Tollens: "Ueber das Allylcyanür oder Crotonitril", in: Justus Liebigs Annalen der Chemie, 1871, 159 (1), S. 105–109; doi:10.1002/jlac.18711590110.
- ↑ C. Pomeranz: "Ueber Allylcyanid und Allylsenföl", in: Justus Liebigs Annalen der Chemie, 1906, 351, S. 354–362; doi:10.1002/jlac.19073510127.
- ↑ F. Beilstein: "Handbuch der organischen Chemie", 3. Auflage, 1. Band. Verlag Leopold Voss, 1893. S. 506 (Volltext).
- ↑ S. Shimizu, S. Kekka, S. Kashino, M. Haisa: "Topochemical Studies. III. The Crystal and Molecular Structures of Crotonic Acid, CH3CH=CHCOOH, and Crotonamide, CH3CH=CHCONH2", in: Bulletin of the Chemical Society of Japan, 1974, 47 (7), S. 1627–1631.
- 1 2 Eintrag zu Butensäure. In: Römpp Online. Georg Thieme Verlag, abgerufen am 10. März 2014.
- 1 2 3 4 Heilbron: Dictionary of organic compounds, Volume One, 1953, S. 615 (Fulltext).
- ↑ J. M. Lovén, H. Johansson: "Einige schwefelhaltige β-Substitutionsderivate der Buttersäure", in: Berichte der deutschen chemischen Gesellschaft, 1915, 48 (2), S. 1254–1262; doi:10.1002/cber.19150480205.
- ↑ A. M. Clover, G. F. Richmond: "The Hydrolysis of Organic Peroxides and Peracids", in: American Chemical Journal, 1903, 29 (3); S. 179–203 (Fulltext).
- ↑ F. Beilstein: "Handbuch der organischen Chemie", 3. Auflage, 1. Band. Verlag Leopold Voss, 1893. S. 562 (Volltext).
- ↑ Carter, H. E.; West, H. D. (1955). "dl-Threonine". Org. Synth.; Coll. Vol., 3, p. 813
- ↑ A. Kleemann u. J. Engel: Pharmazeutische Wirkstoffe: Synthesen, Patente, Anwendungen, 2. neubearb. u. erw. Aufl., Band 5, Georg Thieme Verlag Stuttgart, New York, ISBN 3-13-558402-X, S. 251.