Dithionite
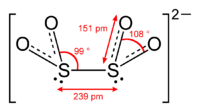
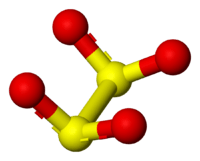
The dithionite anion ([S2O4]2−), is an oxoanion of sulfur [1] formally derived from dithionous acid, H2S2O4.
Chemistry
Dithionous acid has not been detected either as a pure compound or in solution. Dithionite ions undergo both acid and alkaline hydrolysis to thiosulfate and bisulfite, and sulfite and sulfide, respectively
- 2 S2O2−
4 + H2O → S2O2−
3 + 2 HSO−
3
- 3 Na2S2O4 + 6 NaOH → 5 Na2SO3 + Na2S + 3 H2O
Uses
The sodium salt of dithionous acid, sodium dithionite, finds widespread use in industry as a reducing agent.
Dithionite is often used in conjunction with complexing agent (e.g., citric acid) to reduce iron(III) oxy-hydroxide into soluble iron(II) compounds and to remove amorphous iron(III)-bearing mineral phases in soil analyses (selective extraction).
The decomposition of dithionite produces reduced species of sulfur that can be very aggressive for the corrosion of steel and stainless steel. Thiosulfate (S2O2−
3) is well known to induce pitting corrosion while dissolved free sulfide (Na2S) is responsible for stress corrosion cracking (SCC).
References
- ↑ International Union of Pure and Applied Chemistry (2005). Nomenclature of Inorganic Chemistry (IUPAC Recommendations 2005). Cambridge (UK): RSC–IUPAC. ISBN 0-85404-438-8. p. 130. Electronic version.
- Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 0-08-037941-9.
| Wikimedia Commons has media related to Dithionites. |