Estratetraenol
 | |
| Clinical data | |
|---|---|
| ATC code | none |
| Identifiers | |
| |
| CAS Number |
1150-90-9 |
| PubChem (CID) | 101988 |
| ChemSpider | 92135 |
| ChEBI | CHEBI:62849 |
| Chemical and physical data | |
| Formula | C18H22O |
| Molar mass | 254.366 g/mol |
| 3D model (Jmol) | Interactive image |
| |
| |
| (verify) | |
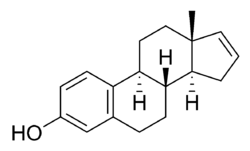
Estratetraenol, also known as estra-1,3,5(10),16-tetraen-3-ol, is an endogenous steroid found in women[1] that has been described as having pheromone-like activities in primates,[2] including humans.[3][4][5][6] Estratetraenol is synthesized from androstadienone by aromatase likely in the ovaries,[7] and is related to the estrogen sex hormones, yet has no known estrogenic effects. It was first identified from the urine of pregnant women.[8]
Estratetraenyl acetate, or estra-1,3,5(10),16-tetraen-3-yl acetate, is a more potent synthetic derivative of estratetraenol.[9]
Estratetraenol is an estrane (C18) steroid and an analogue of estradiol where the C17β hydroxyl group has been removed and a double bond has been formed between the C16 and C17 positions.
See also
References
- ↑ Thysen, B; Elliott, WH; Katzman, PA (1968). "Identification of estra-1,3,5(10),16-tetraen-3-ol (estratetraenol) from the urine of pregnant women (1)". Steroids. 11 (1): 73–87. doi:10.1016/S0039-128X(68)80052-2. PMID 4295975.
- ↑ Laska, M; Wieser, A; Salazar, LT (2006). "Sex-specific differences in olfactory sensitivity for putative human pheromones in nonhuman primates". Journal of Comparative Psychology. 120 (2): 106–12. doi:10.1037/0735-7036.120.2.106. PMID 16719588.
- ↑ Jacob, S; Hayreh, DJ; McClintock, MK (2001). "Context-dependent effects of steroid chemosignals on human physiology and mood". Physiology & Behavior. 74 (1–2): 15–27. doi:10.1016/S0031-9384(01)00537-6. PMID 11564447.
- ↑ Savic, I; Berglund, H; Gulyas, B; Roland, P (2001). "Smelling of odorous sex hormone-like compounds causes sex-differentiated hypothalamic activations in humans". Neuron. 31 (4): 661–8. doi:10.1016/S0896-6273(01)00390-7. PMID 11545724.
- ↑ Berglund, H; Lindström, P; Savic, I (2006). "Brain response to putative pheromones in lesbian women". Proceedings of the National Academy of Sciences of the United States of America. 103 (21): 8269–74. doi:10.1073/pnas.0600331103. PMC 1570103
 . PMID 16705035.
. PMID 16705035. - ↑ Berglund, H; Lindström, P; Dhejne-Helmy, C; Savic, I (2008). "Male-to-female transsexuals show sex-atypical hypothalamus activation when smelling odorous steroids". Cerebral cortex (New York, N.Y. : 1991). 18 (8): 1900–8. doi:10.1093/cercor/bhm216. PMID 18056697.
- ↑ Joseph Johannes; Agnes Maria Weusten (1989). Biochemical pathways in human testicular steroidogenesis (PDF). Pressa Trajectina.
- ↑ Thysen B, Elliott WH, Katzman PA (1968). "Identification of estra-1,3,5(10),16-tetraen-3-ol (estratetraenol) from the urine of pregnant women (1)". Steroids. 11 (1): 73–87. doi:10.1016/s0039-128x(68)80052-2. PMID 4295975.
- ↑ Lundström (2005). Human pheromones : psychological and neurological modulation of a putative human pheromone. Uppsala: Acta Universitatis Upsaliensis. p. 17. ISBN 91-554-6297-9.