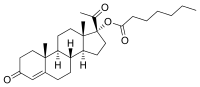
Hydroxyprogesterone heptanoate
 | |
| Clinical data | |
|---|---|
| ATC code | G03DA03 (WHO) |
| Identifiers | |
| |
| Synonyms | 17α-Hydroxypregn-4-ene-3,20-dione 17α-heptanoate |
| CAS Number | 4596-16-1 |
| PubChem (CID) | 94296 |
| ChemSpider | 85099 |
| Chemical and physical data | |
| Formula | C28H42O4 |
| Molar mass | 442.63068 g/mol |
| 3D model (Jmol) | Interactive image |
| |
| |
Hydroxyprogesterone heptanoate (brand names H.O.P., Lutogil A.P., Lutogyl A.P.), or 17α-hydroxyprogesterone heptanoate, also known as hydroxyprogesterone enanthate,[1] is a steroidal progestin of the 17α-hydroxyprogesterone group that was developed by Roussel and Théramex and has been used clinically in France and Monaco.[2][3][4][5] It was a component of the experimental preparation Trophoboline (or Trophobolene), which also contained nandrolone decanoate and estrapronicate.[6][7][8] It was also studied or used in Tocogestan, a combination of hydroxyprogesterone heptanoate (200 mg), progesterone (50 mg), and tocopherol (250 mg) oil for intramuscular injection.[9][5][10]
See also
References
- ↑ Batres, E.; Gomez, R.; Rosenkranz, G.; Sondheimer, F. (1956). "Notes - Steroids. LXXVI. Synthesis of Long Chain Carboxylic Acid Esters of 17α-Hydroxyprogesterone". The Journal of Organic Chemistry. 21 (2): 240–241. doi:10.1021/jo01108a601. ISSN 0022-3263.
- ↑ J. Elks (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 665–. ISBN 978-1-4757-2085-3.
- ↑ Index Nominum 2000: International Drug Directory. Taylor & Francis. January 2000. pp. 532–. ISBN 978-3-88763-075-1.
- ↑ Muller (19 June 1998). European Drug Index: European Drug Registrations, Fourth Edition. CRC Press. pp. 612–. ISBN 978-3-7692-2114-5.
- 1 2 Axel Kleemann; Jürgen Engel (2001). Pharmaceutical Substances: Syntheses, Patents, Applications. Thieme. p. 1033. ISBN 978-3-13-558404-1.
- ↑ Excerpta medica. Section 8, Neurology and neurosurgery. 1981. p. 10.
- ↑ Testosterone Congeners—Advances in Research and Application: 2013 Edition: ScholarlyBrief. ScholarlyEditions. 21 June 2013. pp. 137–. ISBN 978-1-4816-9288-5.
- ↑ Alberto Frigerio (1981). Chromatography in Biochemistry, Medicine and Environmental Research: Proceedings of the ... International Symposium on Chromatography in Biochemistry, Medicine and Environmental Research. Elsevier Scientific Publishing Company. p. 99.
- ↑ https://www.google.com/patents/US6774122
- ↑ http://content.iospress.com/articles/international-journal-of-risk-and-safety-in-medicine/jrs132
This article is issued from Wikipedia - version of the 11/5/2016. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.