Molindone
 | |
| Clinical data | |
|---|---|
| Pronunciation | moe-LIN-done |
| Trade names | Moban |
| AHFS/Drugs.com | Consumer Drug Information |
| MedlinePlus | a682238 |
| Pregnancy category |
|
| Routes of administration | By mouth (tablets) |
| ATC code | N05AE02 (WHO) |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Metabolism | Hepatic |
| Biological half-life | 1.5 hours |
| Excretion | Minor, renal and fecal |
| Identifiers | |
| |
| CAS Number |
7416-34-4 |
| PubChem (CID) | 23897 |
| IUPHAR/BPS | 207 |
| DrugBank |
DB01618 |
| ChemSpider |
22342 |
| UNII |
RT3Y3QMF8N |
| KEGG |
D08226 |
| ChEMBL |
CHEMBL460 |
| Chemical and physical data | |
| Formula | C16H24N2O2 |
| Molar mass | 276.374 g/mol |
| 3D model (Jmol) | Interactive image |
| |
| |
| | |
Molindone (Moban) is a therapeutic antipsychotic, used in the treatment of schizophrenia.[1] It works by blocking the effects of dopamine in the brain, leading to diminished psychoses. It is rapidly absorbed when taken orally.
It is sometimes described as a typical antipsychotic,[2] and sometimes described as an atypical antipsychotic.[3]
Molindone was discontinued by its previous supplier, Endo Pharmaceuticals, on January 13, 2010.[4] It is currently available in the U.S. from CorePharma under Abbreviated New Drug Application approved March 23, 2015.
Adverse effects
The side effect profile of molindone is similar to that of other typical antipsychotics. Unlike most antipsychotics, however, molindone use is associated with weight loss.[3][5]
Synthesis
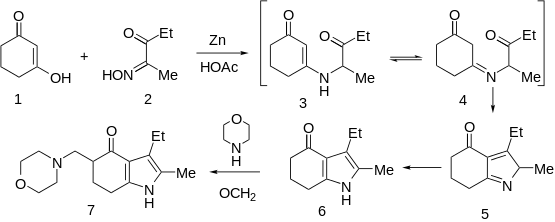
Condensation of oximinoketone 2 (from nitrosation of 3-pentanone), with cyclohexane-1,3-dione (1) in the presence of zinc and acetic acid leads directly to the partly reduced indole derivative 6. The transformation may be rationalized by assuming as the first step, reduction of 2 to the corresponding α-aminoketone. Conjugate addition of the amine to 1 followed by elimination of hydroxide (as water) would give ene-aminoketone 3. This enamine may be assumed to be in tautomeric equilibrium with imine 4. Aldol condensation of the side chain carbonyl group with the doubly activated ring methylene group would then result in cyclization to pyrrole 5; simple tautomeric transformation would then give the observed product. Mannich reaction of 6 with formaldehyde and morpholine gives the tranquilizer molindone (7).
See also
References
- ↑ "molindone". F.A. Davis Company.
- ↑ Aparasu RR, Jano E, Johnson ML, Chen H (October 2008). "Hospitalization risk associated with typical and atypical antipsychotic use in community-dwelling elderly patients". Am J Geriatr Pharmacother. 6 (4): 198–204. doi:10.1016/j.amjopharm.2008.10.003. PMID 19028375.
- 1 2 Bagnall A, Fenton M, Kleijnen J, Lewis R (2007). Bagnall, Anne-Marie, ed. "Molindone for schizophrenia and severe mental illness". Cochrane Database Syst Rev (1): CD002083. doi:10.1002/14651858.CD002083.pub2. PMID 17253473.
- ↑ http://www.fda.gov/Drugs/DrugSafety/DrugShortages/ucm050794.htm
- ↑ Allison DB, Mentore JL, Heo M, et al. (1999). "Antipsychotic-induced weight gain: a comprehensive research synthesis". Am J Psychiatry. 156 (11): 1686–96. PMID 10553730. Free full text