Aureusidin synthase
| Aureusidin synthase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
| EC number | 1.21.3.6 | ||||||||
| CAS number | 320784-48-3 | ||||||||
| Databases | |||||||||
| IntEnz | IntEnz view | ||||||||
| BRENDA | BRENDA entry | ||||||||
| ExPASy | NiceZyme view | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| |||||||||
Aureusidin synthase (EC 1.21.3.6, AmAS1) is an enzyme with systematic name 2',4,4',6'-tetrahydroxychalcone 4'-O-beta-D-glucoside:oxygen oxidoreductase.[1][2][3][4]
Aureusidin synthase has two main enzymatic tasks: hydroxylation at the 3-position on the B-ring of chalcones, and the oxidative cyclization of chalcones to form aurones.[2] The chalcones modified are typically glucosylated 2',4,4',6'-tetrahydroxychalcone (THC) and 2',3,4,4',6'-pentahydroxychalcone (PHC).[2] These aurones, particularly auresidin, form pigments for coloration in flowers.[1] These pigments may have been developed to attract and guide bees for pollination,[5] but they also provide protection from viruses, pests and fungus.[6]
Enzyme structure
Aureusidin synthase is a 39 kDa monomeric glycoprotein containing binuclear copper.[1] The addition of phenylthiourea, which competitively binds to binuclear copper,[7] inhibits the enzyme's productivity overall.[2] Because of this, it is likely that the active site contains the binuclear copper.
Aureusidin synthase is homologous to plant polyphenol oxidase (PPO),[1] but contains certain significant modifications. While PPO has a highly conserved N-terminal amino acid sequence in order to facilitate transport to the plastid lumen, aureusidin synthase lacks this sequence[1] and thus is not localized to the plasmid. Much like PPOs,[8] aureusidin synthase is likely first synthesized as a larger ~60 kDa protein and then undergoes proteolytic cleavage to remove transport groups.[1]
Enzyme mechanism
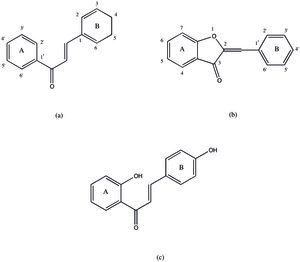
Aureusidin synthase catalyzes the creation of aurones from chalcones through hydroxylation and oxidative cyclization. This class of reactions includes:
- (1) 2',4,4',6'-tetrahydroxychalcone 4'-O-beta-D-glucoside + O2 aureusidin 6-O-beta-D-glucoside + H2O
- (2) 2',3,4,4',6'-pentahydroxychalcone 4'-O-beta-D-glucoside + 1/2 O2 aureusidin 6-O-beta-D-glucoside + H2O
- (3) 2',3,4,4',6'-pentahydroxychalcone 4'-O-beta-D-glucoside + O2 bracteatin 6-O-beta-D-glucoside + H2O
While the protein is named for the yellow aureusidin pigment it often produces, it may produce a number of similar aurones including sulfuretin, bracteatin, and 3',4',5',6-tetrahydroxyaurone. In order to modify the chalcone to an aurone, the chalcone must undergo an oxidative cyclization to form a five-member heterocycle fused to the a-ring of the aurone. This step may not proceed unless the 3-position on the chalcone's B-ring is oxygenated. From this and the protein's homology with PPO, the current proposed mechanism for aureusidin synthase is shown in Fig. 2. The first step in the process results in a hydroxylation in the 3 position (for THC) and an oxidation to a diketone ring. This mechanism is generally assumed to be the same as that of tyrosinase.[9] This assumption is based on the homology of the two structures and similar functionality. This product then undergoes an oxidative cyclization and then an isomerization to give the final product, but it is possible that these steps take place outside the enzyme.[2] This mechanism can only produce aurones with 3',4'-dihydroxy or 3',4',5'-trihydroxy functionalization, but there have been aurones reported with one or no hydroxyl groups on the B-ring. It is unclear whether this aurone formation is due to an alternative mechanism or another protein.[10]

Biological function
Aureusidin is a plant flavonoid that provides yellow coloration in several plants, including snapdragons and cosmos.[1] It also provides various protective benefits from disease and parasites.[6] Auresidin synthase is active only in the vacuole, and it is transported directly from the endoplasmic reticulum to the vacuole via Golgi body.[4] In vitro studies have shown that aurone synthesis proceeds much more quickly when the chalcone is first glucosylated,[2] and in vivo studies have shown that yellow coloration is not expressed without the coexpression of the UDP glucuronosyltransferase UGT88D3 to first glucosylate the chalcone.[4] This glucosylation aids in aurone production by metabolically channelling the modified chalcones to the vacuole.[11]
Industrial relevance
The genetic modification of flowering plants to express colors not possible by natural breeding has been one of the main goals of the floricultural industry.[12] Aureusidin synthase has been introduced into plants with naturally blue flowers to form new transgenic yellow flowers in Torenia species.[4] This is accomplished by coexpressing Aureusidin synthase and UDP-glucose:chalcone 4'-O-glucosyltransferase in flowers and using RNA interference to block the expression of natural pigment production.[4]
In addition to its importance to the floricultural industry, aureusidin synthase is a relatively new target for biomedical research.[13] Aurones have been found to have antioxidant, antibacterial, and anticancer effects.[14] Aureusidin synthase is of particular attractiveness in creating genetically modified plants that may confer medicinal properties, and the creation of transgenic lettuce and tobacco leaves with improved antioxidant properties has already been demonstrated.[15]
References
- 1 2 3 4 5 6 7 Nakayama, T; Yonekura-Sakakibara, K; Sato, T; Kikuchi, S; Fukui, Y; Fukuchi-Mizutani, M; Ueda, T; Nakao, M; Tanaka, Y; Kusumi, T; Nishino, T (2000). "Aureusidin synthase: A polyphenol oxidase homolog responsible for flower coloration". Science. 290 (5494): 1163–6. doi:10.1126/science.290.5494.1163. PMID 11073455.
- 1 2 3 4 5 6 Nakayama, T; Sato, T; Fukui, Y; Yonekura-Sakakibara, K; Hayashi, H; Tanaka, Y; Kusumi, T; Nishino, T (2001). "Specificity analysis and mechanism of aurone synthesis catalyzed by aureusidin synthase, a polyphenol oxidase homolog responsible for flower coloration". FEBS Letters. 499 (1–2): 107–11. doi:10.1016/S0014-5793(01)02529-7. PMID 11418122.
- ↑ Sato, T; Nakayama, T; Kikuchi, S; Fukui, Y; Yonekura-Sakakibara, K; Ueda, T; Nishino, T; Tanaka, Y; Kusumi, T (2001). "Enzymatic formation of aurones in the extracts of yellow snapdragon flowers". Plant science : an international journal of experimental plant biology. 160 (2): 229–236. doi:10.1016/s0168-9452(00)00385-x. PMID 11164594.
- 1 2 3 4 5 Ono, E; Fukuchi-Mizutani, M; Nakamura, N; Fukui, Y; Yonekura-Sakakibara, K; Yamaguchi, M; Nakayama, T; Tanaka, T; Kusumi, T; Tanaka, Y (2006). "Yellow flowers generated by expression of the aurone biosynthetic pathway". Proceedings of the National Academy of Sciences. 103 (29): 11075–80. doi:10.1073/pnas.0604246103. PMC 1544175
 . PMID 16832053.
. PMID 16832053. - ↑ Lunau, K.; Wacht, S.; Chittka, L. (1996). "Colour choices of naive bumble bees and their implications for colour perception". Journal of Comparative Physiology A. 178 (4). doi:10.1007/BF00190178.
- 1 2 Boumendjel, A (2003). "Aurones: A subclass of flavones with promising biological potential". Current medicinal chemistry. 10 (23): 2621–30. doi:10.2174/0929867033456468. PMID 14529476.
- ↑ Klabunde, T.; Eicken, C.; Sacchettini, J. C.; Krebs, B. (1998). "Crystal structure of a plant catechol oxidase containing a dicopper center". Nature Structural Biology. 5 (12): 1084–1090. doi:10.1038/4193.
- ↑ Robinson, S. P.; Dry, I. (May 1992). "Broad Bean Leaf Polyphenol Oxidase Is a 60-Kilodalton Protein Susceptible to Proteolytic Cleavage". Plant Physiology. 99 (1): 317–323. doi:10.1104/pp.99.1.317.
- ↑ Sánchez-Ferrer, A; Rodríguez-López, J. N.; García-Cánovas, F; García-Carmona, F (1995). "Tyrosinase: A comprehensive review of its mechanism". Biochimica et Biophysica Acta. 1247 (1): 1–11. doi:10.1016/0167-4838(94)00204-t. PMID 7873577.
- ↑ Rathmell, W. G.; Bendall, D. S. (1972). "The peroxidase-catalysed oxidation of a chalcone and its possible physiological significance". The Biochemical Journal. 127 (1): 125–32. doi:10.1042/bj1270125. PMC 1178567
 . PMID 5073737.
. PMID 5073737. - ↑ Jørgensen, K.; Rasmussen, A. V.; Morant, M.; Nielsen, A. H.; Bjarnholt, N.; Zagrobelny, M.; Bak, S. R.; Møller, B. L. (2005). "Metabolon formation and metabolic channeling in the biosynthesis of plant natural products". Current Opinion in Plant Biology. 8 (3): 280–291. doi:10.1016/j.pbi.2005.03.014.
- ↑ Mol, J.; Cornish, E.; Mason, J.; Koes, R. (1999). "Novel coloured flowers". Current Opinion in Biotechnology. 10 (2): 198–201. doi:10.1016/S0958-1669(99)80035-4.
- ↑ Haudecoeur, R; Boumendjel, A (2012). "Recent advances in the medicinal chemistry of aurones". Current medicinal chemistry. 19 (18): 2861–75. doi:10.2174/092986712800672085. PMID 22519399.
- ↑ Detsi, A.; Majdalani, M.; Kontogiorgis, C. A.; Hadjipavlou-Litina, D.; Kefalas, P. (2009). "Natural and synthetic 2′-hydroxy-chalcones and aurones: Synthesis, characterization and evaluation of the antioxidant and soybean lipoxygenase inhibitory activity". Bioorganic & Medicinal Chemistry. 17 (23): 8073–8085. doi:10.1016/j.bmc.2009.10.002.
- ↑ Shakya, R; Ye, J; Rommens, C. M. (2012). "Altered leaf colour is associated with increased superoxide-scavenging activity in aureusidin-producing transgenic plants". Plant Biotechnology Journal. 10 (9): 1046–55. doi:10.1111/j.1467-7652.2012.00732.x. PMID 22924954.
External links
- Aureusidin synthase at the US National Library of Medicine Medical Subject Headings (MeSH)
