Iron(II) fluoride
 | |
| Identifiers | |
|---|---|
| 7789-28-8 anhydrous 13940-89-1 (tetrahydrate) | |
| 3D model (Jmol) | Interactive image |
| ChemSpider | 74215 |
| ECHA InfoCard | 100.029.232 |
| PubChem | 522690 |
| |
| |
| Properties | |
| FeF2 | |
| Molar mass | 93.84 g/mol (anhydrous) 165.902 g/mol (tetrahydrate) |
| Appearance | colorless transparent crystals[1] |
| Density | 4.09 g/cm3 (anhydrous) 2.20 g/cm3 (tetrahydrate) |
| Melting point | 970 °C (1,780 °F; 1,240 K) (anhydrous) 100 °C (tetrahydrate)[2] |
| Boiling point | 1,100 °C (2,010 °F; 1,370 K) (anhydrous) |
| 165 g/100 mL | |
| Solubility | insoluble in ethanol, ether; dissolves in HF |
| Structure | |
| Rutile (tetragonal), tP6 | |
| P42/mnm, No. 136 | |
| Hazards | |
| Main hazards | Causes severe skin burns & eye damage; Hazardous decomposition products formed under fire conditions- Iron oxides[3] |
| Flash point | not applicable[3] |
| Related compounds | |
| Other anions |
iron(II) oxide, iron(II) chloride |
| Other cations |
manganese(II) fluoride, cobalt(II) fluoride |
| Related compounds |
iron(III) fluoride |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Iron(II) fluoride or ferrous fluoride is an inorganic compound with the molecular formula FeF2. It forms a tetrahydrate FeF2·4H2O that is often referred to by the same names. The anhydrous and hydrated forms are white crystalline solids.[1][4]
Structure and bonding
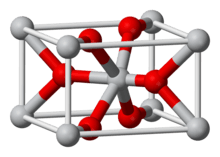
Anhydrous FeF2 adopts the TiO2 rutile structure. As such, the iron cations are octahedral and fluoride anions are trigonal planar.[5][6]
The tetrahydrate can exist in two structures, or polymorphs. One form is rhombohedral and the other is hexagonal, the former having a disorder.[1]
Like most fluoride compounds, the anhydrous and hydrated forms of iron(II) fluoride feature high spin metal center. Low temperature neutron diffraction studies show that the FeF2 is antiferromagnetic.[7] Heat capacity measurements reveal an event at 78.3 K corresponding to ordering of antiferromagnetic state.[8]
Selected physical properties
FeF2 sublimes between between 958 and 1178 K. Using Torsion and Knudsen methods, the heat of sublimation was experimentally determined and averaged to be 271 ± 2 kJ mole−1.[9]
The following reaction is proposed in order to calculate the atomization energy for Fe+:[10]
- FeF2 + e → Fe+ + F2 (or 2F) + 2e
Synthesis and reactions
The anhydrous salt can be prepared by reaction of ferrous chloride with anhydrous hydrogen fluoride.[11] It is slightly soluble in water (with solubility product Ksp = 2.36×10−6 at 25 °C)[12] as well as dilute hydrofluoric acid, giving a pale green solution.[1] It is insoluble in organic solvents.[4]
The tetrahydrate can be prepared by dissolving iron in warm hydrated hydrofluoric acid and precipitating the result by addition of ethanol.[1] It oxidizes in moist air to give, inter alia, a hydrate of iron(III) fluoride, (FeF3)2·9H2O.[1]
Uses
FeF2 is used to catalyze some organic reactions.[13]
External links
References
- 1 2 3 4 5 6 Penfold, B. R.; Taylor, M. R. (1960). "The crystal structure of a disordered form of iron(II) fluoride tetrahydrate". Acta Crystallographica. 13: 953–956. doi:10.1107/S0365110X60002302.
- ↑ Pradyot Patnaik. Handbook of Inorganic Chemicals. McGraw-Hill, 2002, ISBN 0-07-049439-8
- 1 2 Sigma-Aldrich. "Material Safety Data Sheet". Sigma-Aldrich. Retrieved 5 April 2011.
- 1 2 Dale L. Perry (1995), "Handbook of Inorganic Compounds", page 167. CRC Press. ISBN 9780849386718
- ↑ Stout, J.; Stanley A. Reed (1954). "The Crystal Structure of MnF2, FeF2, CoF2, NiF2 and ZnF2". J. Am. Chem. Soc. 76: 5279–5281. doi:10.1021/ja01650a005.
- ↑ M.J.M., de Almeida,; M.M.R., Costa,; J.A., Paixão, (1989-12-01). "Charge density of FeF2". Acta Crystallographica Section B: Structural Science. 45 (6). doi:10.1107/S0108768189007664. ISSN 0108-7681.
- ↑ Erickson, R. (June 1953). "Neutron Diffraction Studies of Antiferromagnetism in Manganous Fluoride and Some Isomorphous Compounds". Physical Review. 90 (5): 779–785. doi:10.1103/PhysRev.90.779.
- ↑ Stout, J.; Edward Catalano (December 1953). "Thermal Anomalies Associated with the Antiferromagnetic Ordering of FeF2, CoF3, and NiF2". Physical Review. 92 (6): 1575–1575. doi:10.1103/PhysRev.92.1575.
- ↑ Bardi, Gianpiero; Brunetti, Bruno; Piacente, Vincenzo (1996-01-01). "Vapor Pressure and Standard Enthalpies of Sublimation of Iron Difluoride, Iron Dichloride, and Iron Dibromide". Journal of Chemical & Engineering Data. 41 (1): 14–20. doi:10.1021/je950115w. ISSN 0021-9568.
- ↑ Kent, Richard; John L. Margrave (November 1965). "Mass Spectrometric Studies at High Temperatures. VIII. The Sublimation Pressure of Iron(II) Fluoride". Journal of the American Chemical Society. 87 (21): 4754–4756. doi:10.1021/ja00949a016.
- ↑ W. Kwasnik "Iron(II) Fluoride" in Handbook of Preparative Inorganic Chemistry, 2nd Ed. Edited by G. Brauer, Academic Press, 1963, NY. Vol. 1. p. 266.
- ↑ "SOLUBILITY PRODUCT CONSTANTS" (PDF).
- ↑ Wildermuth, Egon; Stark, Hans; Friedrich, Gabriele; Ebenhöch, Franz Ludwig; Kühborth, Brigitte; Silver, Jack; Rituper, Rafael (2005), "Iron Compounds", Ullmann's Encyclopedia of Industrial Chemistry, Weinheim: Wiley-VCH, doi:10.1002/14356007.a14_591