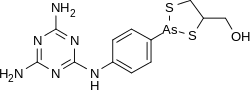
Melarsoprol
 | |
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | Micromedex Detailed Consumer Information |
| Pregnancy category |
|
| Routes of administration | IV |
| ATC code | P01CD01 (WHO) QP51AD04 (WHO) |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | ? |
| Metabolism | ? |
| Biological half-life | 35 hours |
| Excretion | Renal |
| Identifiers | |
| |
| CAS Number |
494-79-1 |
| PubChem (CID) | 10311 |
| ChemSpider |
9889 |
| UNII |
ZF3786Q2E8 |
| KEGG |
D00832 |
| ChEMBL |
CHEMBL166 |
| Chemical and physical data | |
| Formula | C12H15AsN6OS2 |
| Molar mass | 398.341 g/mol |
| 3D model (Jmol) | Interactive image |
| |
| |
| | |
Melarsoprol is a medication used for the treatment of sleeping sickness.[1] It is used in the late or chronic disease states associated with neurological problems. However, it is not used for Chagas' disease. It is given by injection into a vein.
Due to its risk of causing encephalopathy, other medications are first line treatments for stage one of the disease and for certain other trypanosomes. Other common names for this drug include Mel B and Melarsen Oxide Bal.[2] Melarsoprol is the dimercaptopropanol derivative of melarsen oxide (Mel Ox).[3] The active metabolite of Mel B is Mel Ox, which is taken up by the TbAT1 adenosine transporter in T. brucei gambiense or T.b. rhodesiense.[4]
Its effects against late stage trypanosomiasis were first demonstrated by Freidheim in 1949.It is on the World Health Organization's List of Essential Medicines, the most important medications needed in a basic healthcare system.[5] Melarsoprol is currently produced by Sanofi-Aventis and under an agreement with WHO, Sanofi-Aventis donates melarsoprol to countries were the disease is common. Despite production, this medication is not available in Canada and is only available in the United States through the CDC.[6]
Medical uses
People diagnosed with trypanosome-caused disease should be treated with an anti-trypanosomal. Treatment is based on stage, 1 or 2, and parasite,T. b. rhodesiense or T. b. gambiense. In stage 1 disease, trypanosomes are present only in the peripheral circulation. In stage 2 disease, trypanosomes have crossed the blood brain barrier and are present in the central nervous system. [7]
The following are considerable treatment options:[7]
Melarsorprol is a treatment used during the second stage of the disease. So far, it is the only treatment available for late-stage T. b. rhodesiense.[8]
Due to high toxicity, melarsarprol is reserved only for the most dangerous cases. Other agents associated with lower toxicity levels are used during stage 1 of the disease.[1] The approval of the nifurtimox/eflornithine combination therapy (NECT) in 2009 for the treatment of T. b. gambiense limited the use of merlarsoprol in the treatment of second-stage T. b. rhodesiense.[9]
Failure rates of 27% in certain African countries have been reported.[10] This was caused by both drug resistance and additional mechanisms that have not yet been elucidated. Resistance is likely due to transport problems associated with the P2 transporter, an adenine-adenosine transporter. Resistance can occur with point mutations within this transporter.[3] Resitance has been presence since the 1970s.[11]
Side effects
Although melarsoprol cures about 96% of people with late stage disease, its toxicity limits its use.[8] About 1-5% of people die from adverse events related to melarsoprol treatment.[12] As a toxic organic compound of arsenic, melarsoprol is a dangerous treatment that is typically only administered by injection under the supervision of a licensed physician. Notable side effects are similar to arsenic poisoning. Among clinicians, it is colloquially referred to as "arsenic in antifreeze".[13] Severe and life-threatening adverse reactions are associated with melarsoprol. It is known to cause a range of side effects including convulsions, fever, loss of consciousness, rashes, bloody stools, nausea and vomiting. In approximately 5-10% of cases, it causes encephalopathy. Of those, about 50% die due to encephalopathy related adverse reaction.[7] Additional concerns with melarsoprol that may be serious include damage to the heart, presence of albumin in the urine that could be associated with kidney damage and an increase in blood pressure.[12]
Cautions
Numerous warnings have been established before melarsoprol treatment can be initiated. Prior to initiation, the following must be noted: glucose-6-phosphate dehydrogenase deficiency, kidney or liver disease, cardiac problems (high blood pressure, irregular beating of the heart or arrhythmias, any damage to the heart muscles and potential signs of heart failure), preexisting nervous system disorders, and any signs of leprosy.
Routine laboratory testing is needed before and after melarsoprol initiation. Laboratory parameters for both therapeutic effects and toxic effects need to be evaluated.
Blood analysis is used to detect the presence of trypanosomes. An evaluation of the cerebrospinal fluid via a lumbar puncture is also used to determine an individual's white blood count and level of protein. These are diagnostic criteria such that the presence of trypanosomes, an elevated white blood count greater than five per microliter, or a protein content greater than 40mg are considered abnormal and initiation should be considered. Continuous cerebrospinal fluid evaluation should be repeated every six months for at least three years in individuals that have undergone melarsoprol treatment.
To assess potential concerns related to toxicity, the following should be completed: a complete blood count, an assessment of electrolyte levels, liver and kidney function tests and a urinalysis to detect the appearance, concentration and content of the urine.
Pregnancy and breastfeeding
Currently, melarsoprol is not recommended for use in pregnant women. The World Health Organization suggests that treatment be deferred until immediately after delivery since the effects of the medication on the developing fetus have not yet been established.[12]
Lactation guidelines associated with melarsoprol have not yet been established.
Mechanism of action
Melarsoprol is a prodrug, which is metabolized to melarsen oxide (Mel Ox) as its active form. Mel Ox is an arsen-oxide that irreversibly binds to vicinal sulfhydryl groups on pyruvate kinase, which disrupts energy production in the parasite. The inability to distinguish between host and parasites renders this drug highly toxic with many side effects.
Mel Ox reacts with trypanothione (a spermidine-glutathione adduct that replaces glutathione in trypanosomes). By binding to this adduct, it forms a melarsen oxide-trypanothione adduct (Mel T) that competitively inhibits trypanothione reductase, effectively killing the parasitic cell.[3]
Dosage
Two arsenic-containing stereoisomers exist in a 3:1 molar ratio. Since melarsarprol is insoluble in water, dosage occurs via a 3.6% propylene glycol intravenous injection.[3] To avoid the risk of injection site reactions, melarsoprol must be given slowly.
Melarsoprol used for the treatment of African trypanosomiasis with CNS involvement is given under a complicated dosing schedule. The dosing schedule for children and adults is 2-3.6 mg/kg/day intravenously for three days, then repeated every seven days for a total of three series. [7] To monitor for relapse, follow-up is recommended every 6 months for at least 2 years. [12]
Pharmacokinetics
The half-life of melarsoprol is less than one hour, but bioassays indicate a 35 hour half-life. This is commonly associated with pharmacologic agents that have active metabolites. One such metabolite, Mel Ox, reaches maximum plasma levels about 15 minutes after melarsoprol injection. Melarsoprol clearance is 21.5 ml/min/kg and the Mel Ox half-life is approximately 3.9 hours.[14]
References
- 1 2 Bisser S, N'Siesi FX, Lejon V, et al. (2007). "Equivalence trial of melarsoprol and nifurtimox monotherapy and combination therapy for the treatment of second-stage Trypanosoma brucei rhodesiense sleeping sickness". J. Infect. Dis. 195 (3): 322–9. doi:10.1086/510534. PMID 17205469.
- ↑ "Melarsoprol Drug Information, Professional". www.drugs.com. Retrieved 2016-11-09.
- 1 2 3 4 Brunton L (2011). "Goodman & Gillman's The Pharmacological Basis of Therapeutics". McGraw Hill Medical: 1427–28.
- ↑ Schweingruber ME (2004). "The Melaminophenyl Arsenicals Melarsoprol and Melarsen Oxide Interfere with Thiamine Metabolism in the Fission Yeast Schizosaccharomyces pombe.". Antimicrobial Agents and Chemotherapy. 48 (9): 3268–3271. doi:10.1128/AAC.
- ↑ "WHO Model List of Essential Medicines" (PDF). World Health Organization. October 2013. Retrieved 22 April 2014.
- ↑ "Melarsoprol Drug Information, Professional". www.drugs.com. Retrieved 2016-11-09.
- 1 2 3 4 CDC (2013). "Disease Control and Prevention: Parasites Africian Trypanosomiasis".
- 1 2 "Trypanosoma brucei gambiense and Trypanosoma brucei rhodesiense (African Trypanosomiasis) - Infectious Disease and Antimicrobial Agents". www.antimicrobe.org. Retrieved 2016-11-17.
- ↑ Farrar J (2014). "Manson's Tropical Diseases: Expert Consult-Online". 23: 616.
- ↑ "Human African trypanosomiasis". Nature Reviews Microbiology. 2: 186–187. March 2004.
- ↑ "The phenomenon of treatment failures in Human African Trypanosomiasis". Tropical Medicine and International Health. 6: 906–914. November 2001.
- 1 2 3 4 "WHO Model Prescribing Information: Drugs Used in Parasitic Diseases - Second Edition: Protozoa: African trypanosomiasis: Melarsoprol". apps.who.int. Retrieved 2016-11-09.
- ↑ Hollingham R (2005). "Curing diseases modern medicine has left behind". New Scientist. 2005 (2482): 40–41.
- ↑ Keiser J., Ericsson O, Burri C (2000). "Investigations of the metabolites of the trypanocidal drug melarsoprol". Clinical Pharmacology. 67: 478–88. doi:10.1067/mcp.2000.105990.