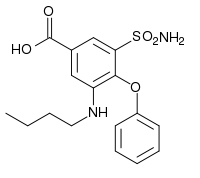
Bumetanide
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Bumex |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a684051 |
| Pregnancy category | |
| Routes of administration | oral, intravenous, intramuscular |
| ATC code | C03CA02 (WHO) |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | almost complete (~80%) |
| Protein binding | 97% |
| Metabolism | hepatic |
| Biological half-life | ~0.8 hours |
| Excretion | renal |
| Identifiers | |
| |
| CAS Number |
28395-03-1 |
| PubChem (CID) | 2471 |
| IUPHAR/BPS | 4837 |
| DrugBank |
DB00887 |
| ChemSpider |
2377 |
| UNII |
0Y2S3XUQ5H |
| KEGG |
D00247 |
| ChEBI |
CHEBI:3213 |
| ChEMBL |
CHEMBL1072 |
| ECHA InfoCard | 100.044.534 |
| Chemical and physical data | |
| Formula | C17H20N2O5S |
| Molar mass | 364.417 g/mol |
| 3D model (Jmol) | Interactive image |
| |
| |
| (verify) | |
Bumetanide (trade names Bumex or Burinex) is a loop diuretic of the sulfamyl category to treat heart failure. It is often used in people in whom high doses of furosemide are ineffective. It is marketed by Hoffmann-La Roche. The main difference between the two substances is in bioavailability and pharmacodynamic potency. Furosemide is incompletely absorbed in the intestine (60%), and there are substantial inter- and intraindividual differences in bioavailability (range 10-90%). Bumetanide is almost completely absorbed (80%), and the absorption is not altered when it is taken with food. It is said to be a more predictable diuretic, meaning that the predictable absorption is reflected in a more predictable effect.[1]
Bumetanide is 40 times more potent than furosemide for patients with normal renal function.[1]
In the brain, bumetanide blocks the NKCC1 cation-chloride co-transporter, and thus decreases internal chloride concentration in neurons. In turn, this concentration change makes the action of GABA more hyperpolarizing, which may be useful for treatment of neonatal seizures, that quite often are not responsive to traditional GABA-targeted treatment, such as barbiturates. Bumetanide is therefore currently under evaluation as a prospective antiepileptic drug.[2]
Abuse
On October 24, 2008, ESPN reported a number of NFL players were being suspended under the steroid policy as a result of taking bumetanide. The drug is often used for weight loss, but also to mask other drugs or steroids by helping to dilute the contents of the user's urine, yielding a lower concentration of filtered substances which may then go undetected.
Bumetanide was an undisclosed active ingredient in the over the counter weight loss supplement StarCaps. StarCaps was pulled off the market after the presence of the undisclosed ingredient was discovered by the Food and Drug Administration.
Synthesis
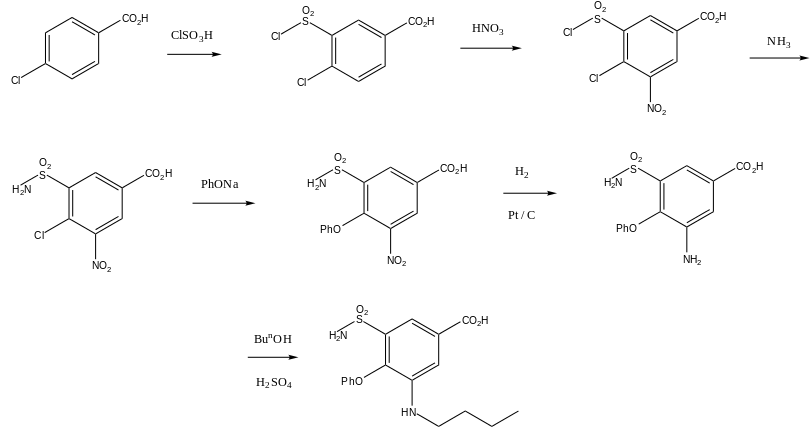
Bumetanide, 3-butylamino-4-phenoxy-5-sulfamoylbenzoic acid, is synthesized from 4-chlorobenzoic acid. In the first stage of synthesis, it undergoes sulfonylchlorination by chlorosulfonic acid, forming 4-chloro-3-chlorosulfonylbenzoic acid, which is further nitrated with nitric acid to 4-chloro-3-chlorosulfonyl-5-nitrobenzoic acid. Reacting this with ammonia gives 5-aminosulfonyl-4-chloro-3-nitrobenzoic acid, which when reacted with sodium phenolate is transformed into 5-amino-sulfonyl-3-nitro-5-phenoxybenzoid acid. Reduction of the nitro group in this product by hydrogen using a palladium on carbon catalyst gives 3-amino-5-aminosulfonyl- 5-phenoxybenzoic acid. Finally, reacting this with butyl alcohol in the presence of sulfuric acid gives the desired bumetanide.
- F.P. Werner, Ger. Pat. DE 1964503 (1970).
- F.P. Werner, Ger. Pat. DE 1964504 (1970).
- P. Feit, U.S. Pat. U.S. Patent 33,806,534 (1974).
- P.W. Feit, U.S. Pat. U.S. Patent 3,634,583 (1972).
- Feit, P. W. (1971). "Aminobenzoic acid diuretics. 2. 4-Substituted-3-amino-5-sulfamoylbenzoic acid derivatives". Journal of Medicinal Chemistry. 14 (5): 432–9. doi:10.1021/jm00287a014. PMID 5117690.
References
- 1 2 Brunton, Laurence; Lazo, John S.; Parker, Keith L., eds. (2006). Goodman & Gilman's The Pharmacological Basis of Therapeutics (11th ed.). New York: McGraw-Hill. pp. 749–753. ISBN 0-07-142280-3.
- ↑ Löscher W, Puskarjov M, Kaila K (June 2013). "Cation-chloride cotransporters NKCC1 and KCC2 as potential targets for novel antiepileptic and antiepileptogenic treatments". Neuropharmacology. 69: 62–74. doi:10.1016/j.neuropharm.2012.05.045. PMID 22705273.
External links
- Bumex (FDA)