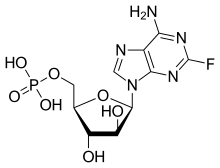
Fludarabine
 | |
| Clinical data | |
|---|---|
| Trade names | Fludara |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a692003 |
| Pregnancy category |
|
| Routes of administration | Intravenous, oral |
| ATC code | L01BB05 (WHO) |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 55% |
| Protein binding | 19 to 29% |
| Biological half-life | 20 hours |
| Excretion | Renal |
| Identifiers | |
| |
| CAS Number |
75607-67-9 |
| PubChem (CID) | 657237 |
| IUPHAR/BPS | 4802 |
| DrugBank |
DB01073 |
| ChemSpider |
571392 |
| UNII |
1X9VK9O1SC |
| KEGG |
D01907 |
| ChEBI |
CHEBI:63599 |
| ChEMBL |
CHEMBL1568 |
| ECHA InfoCard | 100.123.703 |
| Chemical and physical data | |
| Formula | C10H13FN5O7P |
| Molar mass | 365.212 g/mol |
| 3D model (Jmol) | Interactive image |
| |
| |
| | |
Fludarabine or fludarabine phosphate (Fludara) is a chemotherapy drug used in the treatment of hematological malignancies (cancers of blood cells such as leukemias and lymphomas). It is a purine analog, which interferes with DNA synthesis.
It is on the WHO Model List of Essential Medicines, the most important medications needed in a basic health system.[1]
Medical uses
Fludarabine is highly effective in the treatment of chronic lymphocytic leukemia, producing higher response rates than alkylating agents such as chlorambucil alone.[2] Fludarabine is used in various combinations with cyclophosphamide, mitoxantrone, dexamethasone and rituximab in the treatment of indolent non-Hodgkins lymphomas. As part of the FLAG regimen, fludarabine is used together with cytarabine and granulocyte colony-stimulating factor in the treatment of acute myeloid leukaemia. Because of its immunosuppressive effects, fludarabine is also used in some conditioning regimens prior to allogeneic stem cell transplant.
Side effects
Fludarabine is associated with profound lymphopenia, and as a consequence, increases the risk of opportunistic infections significantly. Patients who have been treated with fludarabine will usually be asked to take co-trimoxazole or to use monthly nebulised pentamidine to prevent Pneumocystis jiroveci pneumonia. The profound lymphopenia caused by fludarabine renders patients susceptible to transfusion-associated graft versus host disease, an oftentimes fatal complication of blood transfusion. For this reason, all patients who have ever received fludarabine should only be given irradiated blood components.
Fludarabine causes anemia, thrombocytopenia and neutropenia, requiring regular blood count monitoring. Some patients require blood and platelet transfusion, or G-CSF injections to boost neutrophil counts.
Fludarabine is associated with the development of severe autoimmune hemolytic anemia in a proportion of patients.[3]
Difficulties are often encountered when harvesting peripheral blood stem cells from patients previously treated with fludarabine.[4]
The addition of Fludarabine to medication given for cancer treatment has proved fatal to patients being treated.
Pharmacology
Fludarabine is a purine analog, and can be given both orally and intravenously. Fludarabine inhibits DNA synthesis by interfering with ribonucleotide reductase and DNA polymerase. It is active against both dividing and resting cells. Being phosphorylated, fludarabine is ionized at physiologic pH and is effectually trapped in blood. This provides some level of specificity for blood cells, both cancerous and healthy.
History
Fludarabine was produced by John Montgomery and Kathleen Hewson of the Southern Research Institute in 1968.[5] Their previous work involved 2-fluoroadenosine, which was unsafe for use in humans; the change to this arabinose analogue was inspired by the success of vidarabine.[5] However in June 2016 during a clinical trial of CAR T-cell therapy, two patients died due to swelling of the brain, which occurred after fludarabine was added to the pre-conditioning chemotherapy regimen.
References
- ↑ "19th WHO Model List of Essential Medicines (April 2015)" (PDF). WHO. April 2015. Retrieved May 10, 2015.
- ↑ Rai KR et al. Fludarabine compared with chlorambucil as primary therapy for chronic lymphocytic leukemia. N Engl J Med 2000;343:1750-7. doi:10.1056/NEJM200012143432402 PMID 11114313
- ↑ Gonzalez H et al. Severe autoimmune hemolytic anemia in eight patients treated with fludarabine. Hematol Cell Ther. 1998;40:113-8. PMID 9698219
- ↑ Tournilhac O et al. Impact of frontline fludarabine and cyclophosphamide combined treatment on peripheral blood stem cell mobilization in B-cell chronic lymphocytic leukemia. Blood 2004;103:363-5. PMID 12969985
- 1 2 Sneader, Walter (2005). Drug discovery: a history. New York: Wiley. p. 258. ISBN 0-471-89979-8.
